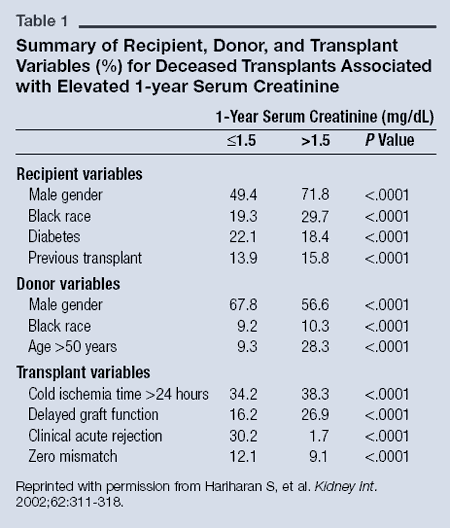
Metformin is a component of a variety of combination products with other anti-diabetic agents. Safety in children has not been determined to this date. The extended-release formulation of metformin is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. 
Metformin is indicated as an adjunct to diet and exercise to improve glycemic control in adults and pediatric patients ≥10 years old with type 2 diabetes mellitus. 23 Type Small Molecule Groups Approved Structure Metformin was first approved in Canada in 1972, 8 and received subsequent FDA approval in the US in 1995. 14 Another well-known benefit of this drug is modest weight loss, making it an effective choice for obese patients type II diabetes. It is commonly described as an "insulin sensitizer", leading to a decrease in insulin resistance and a clinically significant reduction of plasma fasting insulin levels. Metformin is considered an antihyperglycemic drug because it lowers blood glucose concentrations in type II diabetes without causing hypoglycemia. Metformin is a biguanide antihyperglycemic agent and first-line pharmacotherapy used in the management of type II diabetes. Actoplus Met, Avandamet, Fortamet, Glucophage, Glucovance, Glumetza, Glycon, Invokamet, Janumet, Jentadueto, Kazano, Kombiglyze, Komboglyze, Qternmet, Riomet, Segluromet, Synjardy, Trijardy, Velmetia, Xigduo Generic Name Metformin DrugBank Accession Number DB00331 Background
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
